Discovery
Clinical use cases, intended users, cleaning requirements, replacement pressure, and purchasing constraints are captured before product categories are compared.

Getinge helps hospitals, specialty clinics, dental organizations, and care networks compare Dental & Oral Health Equipment, Patient Monitoring & Remote Care, and Consumables & Infection Control Products with clear documentation, service expectations, and product pathways.
Getinge keeps the commercial path visible for teams that need evidence before they can approve a device program.
Clinical use cases, intended users, cleaning requirements, replacement pressure, and purchasing constraints are captured before product categories are compared.
Dental, monitoring, remote care, and infection control options are reviewed against documentation needs, accessories, service coverage, and training expectations.
Value analysis, biomedical engineering, infection prevention, and department leaders receive concise notes that connect claims to records and operating assumptions.
Quotation, implementation handoff, preventive maintenance planning, and consumable continuity are coordinated as one supplier record.
The Getinge workflow keeps sourcing, documentation, service, validation, and hand-off connected for each covered medical device category.
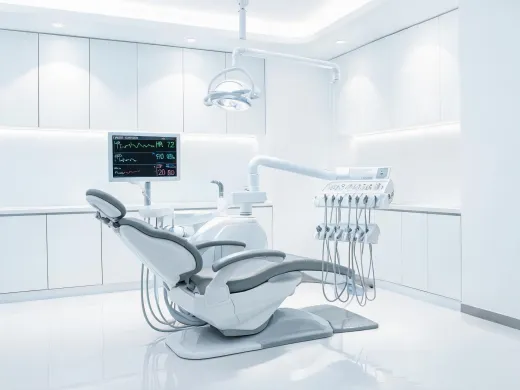
Category reviews include intended care setting, accessories, cleaning workflow, user training, and committee documentation.

Monitoring programs pair device selection with alarm workflow, data handling, cybersecurity notes, and service escalation.

Infection control sourcing is organized around replenishment, lot traceability, reprocessing compatibility, and documented use conditions.

Support records clarify who owns PM cadence, firmware notes, loaners, spare parts, and post-installation training.
"Getinge is useful in committee review because the commercial conversation stays tied to documentation, service ownership, and the lifecycle assumptions our teams have to defend."
Value Analysis Lead, regional hospital networkGetinge's reliable-partner posture is built for teams that need a calm, documented path from category comparison to implementation. Share the care setting, expected quantity, and evidence needs, and the response will be shaped for committee use.